Our technology is revolutionizing radiology. We’d love to work with you to help create better outcomes for patients everywhere.
The first FDA-cleared device that supports concurrent reading, allowing for faster reading with proven, superior, automatic nodule detection.
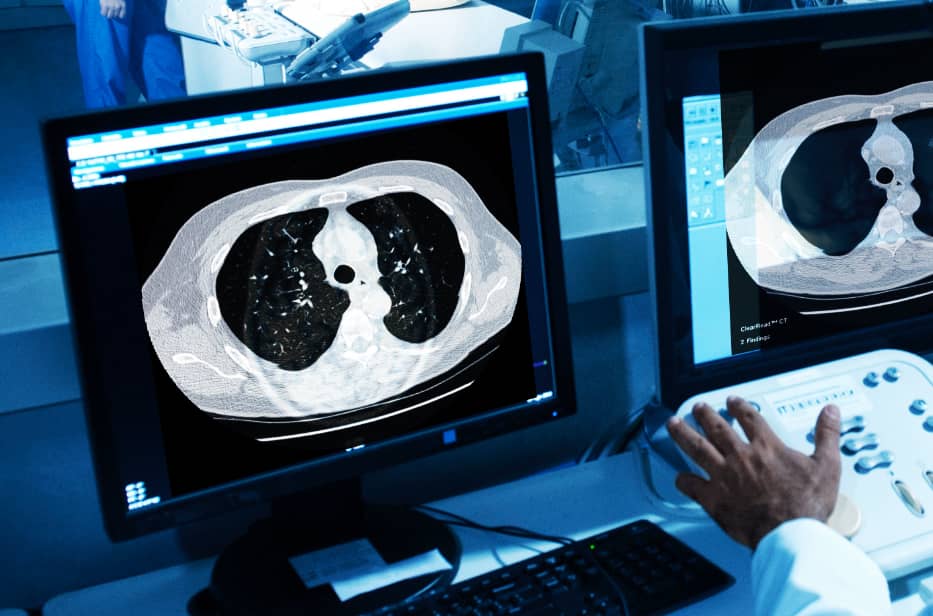
Our five FDA-cleared applications are designed to improve reading efficiency and accuracy across the hospital enterprise without additional equipment, procedures or radiation dose.
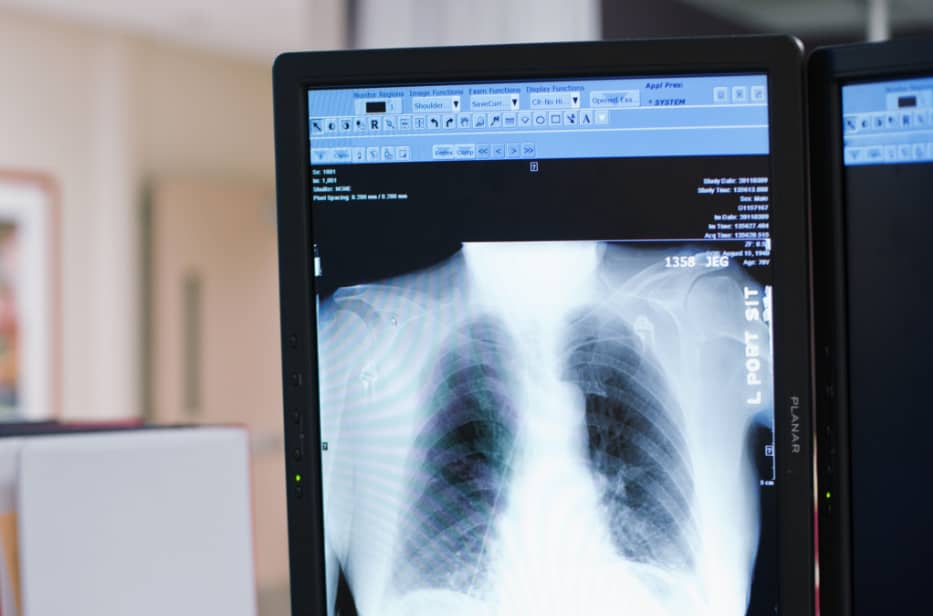
Riverain’s deep learning AI technology is FDA cleared, field tested, and clinically proven to provide real results in radiology.
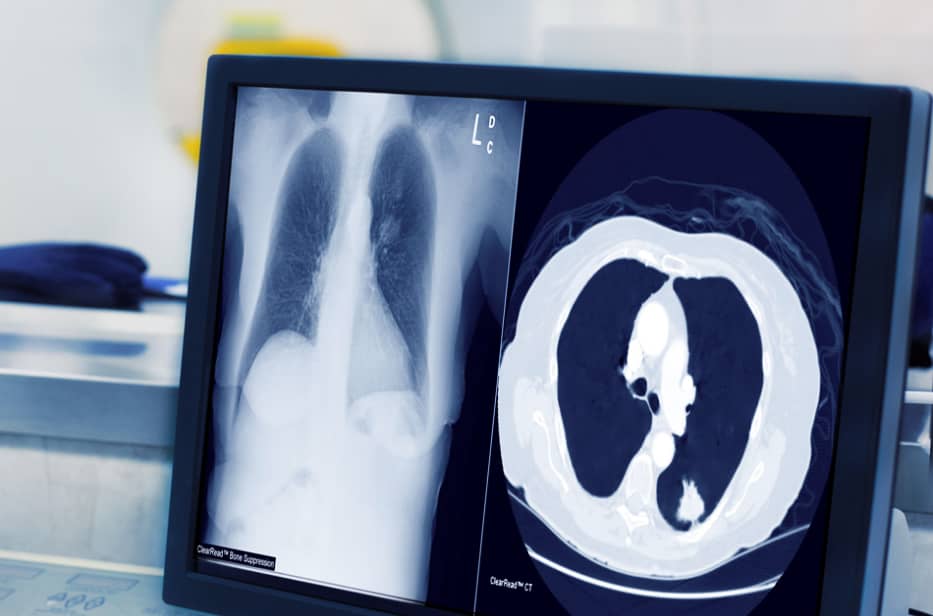
Riverain Technologies advanced AI imaging software is in use by leading healthcare organizations around the world.

Peer reviews, reference accounts, and independent studies confirm that ClearRead technology is positively impacting the field of radiology.
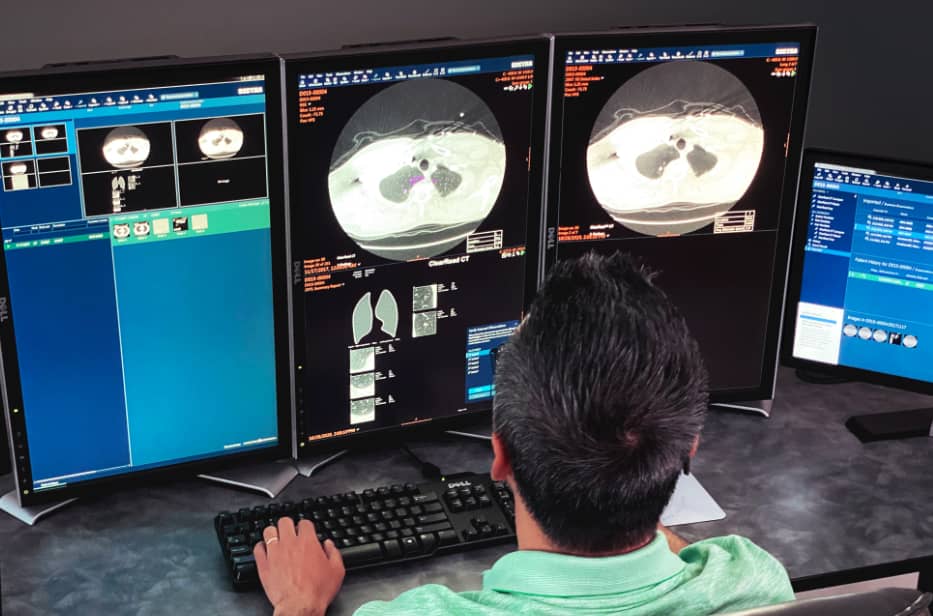
Managing your lung program can seem daunting, whether you’re getting going, growing, or managing workflow. We’ve curated resources for you.
Dayton, Ohio – November 18, 2013 – Licenses sold worldwide through October of this year for Riverain Technologies ClearRead software are up 36 percent compared to the same time period last year.
The sophisticated yet simple-to-use software suppresses the clavicle and ribs that often obscure lung nodules on a conventional X-ray image. Unlike other suppression technology, ClearRead is a software-only solution. There is no additional radiation exposure for patients and no additional equipment, space or staff requirements for the healthcare facility.
“Radiologists are recognizing ClearRead as a readily accessible technology and an instantaneous, cost-effective tool to immediately improve the chest X-ray, one of the most common imaging exams,” said Steve Worrell, CEO of Riverain Technologies. “The software is approved in several countries, and leading healthcare companies are now offering ClearRead products to further enhance their digital radiography systems.”
ClearRead Approved in U.S., Europe and China
All four ClearRead software products are approved and in use in the U.S. and Europe, and ClearRead Bone Suppression™ (formerly SoftView) and +Detect™ (formerly OnGuard) were approved in September 2013 for use in China. The four products are:
“I believe anyone reading chest films needs some type of bone suppression support these days,” said Peter B. Sachs, M.D., Section Chief of Thoracic Imaging and Vice Chair of Informatics, University of Colorado Hospital (UCH) and Anschutz Medical Campus, who has been using ClearRead software since 2011 and sees it as an essential part of his daily workflow.
Because it makes lung nodules more conspicuous, ClearRead Bone Suppression also is reducing the time it takes to read and interpret some X-ray images, he added.
“I can actually spend a little less time looking at the conventional image, or trying to reason through difficult areas on the conventional image,” Dr. Sachs said. “If I think there might be a lesion hiding behind a rib I can go to the bone suppression and, most of the time, feel comfortable that I have an answer.”
Philips, Siemens Offering ClearRead Software
ClearRead software is being offered by leading global healthcare companies as an enhancement to their digital X-ray technology. As of May of this year, Philips Healthcare began offering Riverain Technologies ClearRead applications to its customers with its existing X-ray product line. The relationship expands Riverain’s existing distribution channels, which include Siemens Healthcare, Toyo International and select U.S. distributors.
“Philips offers Riverain products as third party optional items to Philips X-ray solutions,” said Georg Kornweibel, Marketing Diagnostic X-ray, Philips. “Riverain’s ClearRead technology with advanced bone suppression algorithms is a perfect complement to Philips leading digital X-ray systems. Together we offer solutions that contribute to earlier detection of lung nodules and help improve patients’ lives.”
Our technology is revolutionizing radiology. We’d love to work with you to help create better outcomes for patients everywhere.
Subscribe to Riverain via Email
Stay up to date on the latest advancements
Riverain Technologies
3130 South Tech Blvd., Miamisburg, OH 45342
800-990-3387
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |