Our technology is revolutionizing radiology. We’d love to work with you to help create better outcomes for patients everywhere.
The first FDA-cleared device that supports concurrent reading, allowing for faster reading with proven, superior, automatic nodule detection.
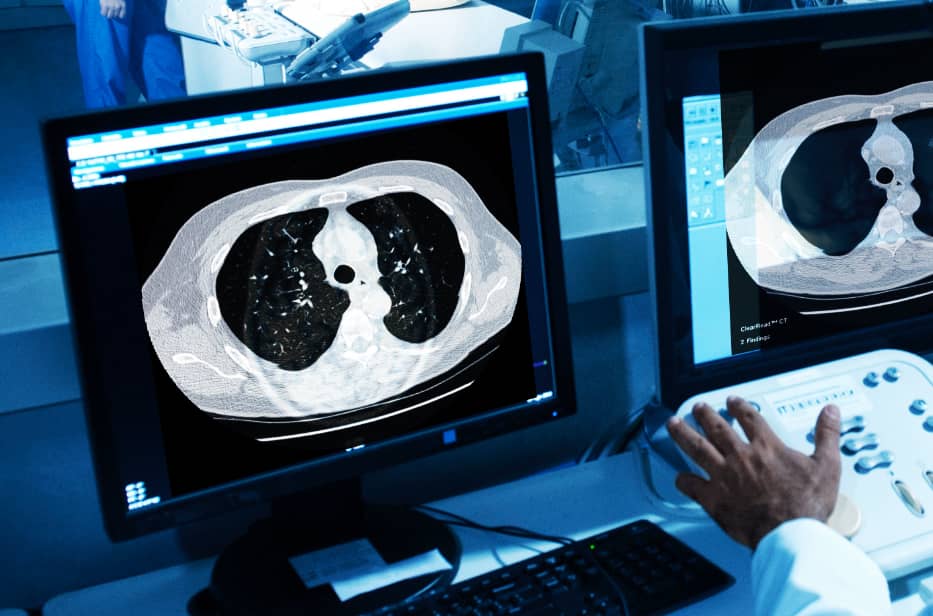
Our five FDA-cleared applications are designed to improve reading efficiency and accuracy across the hospital enterprise without additional equipment, procedures or radiation dose.
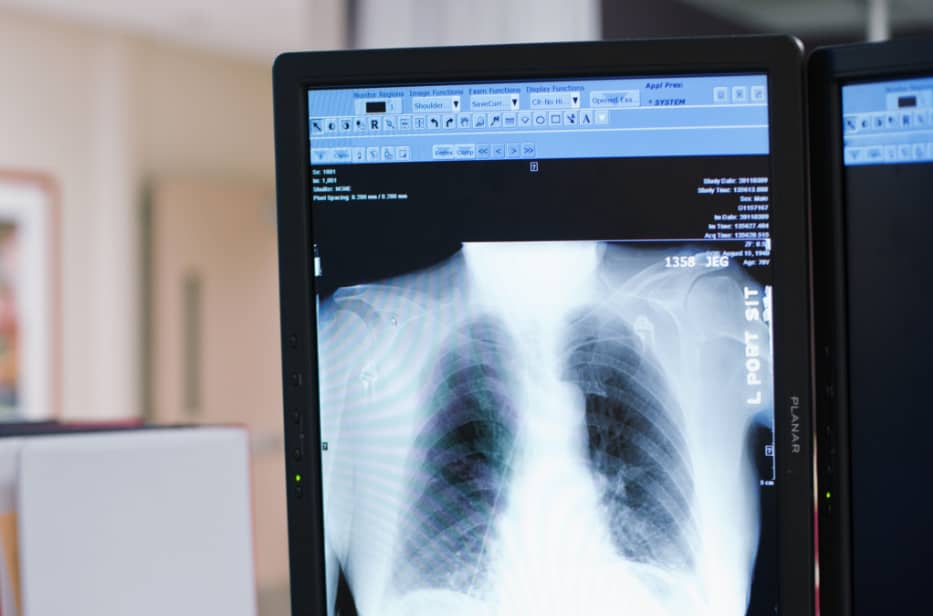
Riverain’s deep learning AI technology is FDA cleared, field tested, and clinically proven to provide real results in radiology.
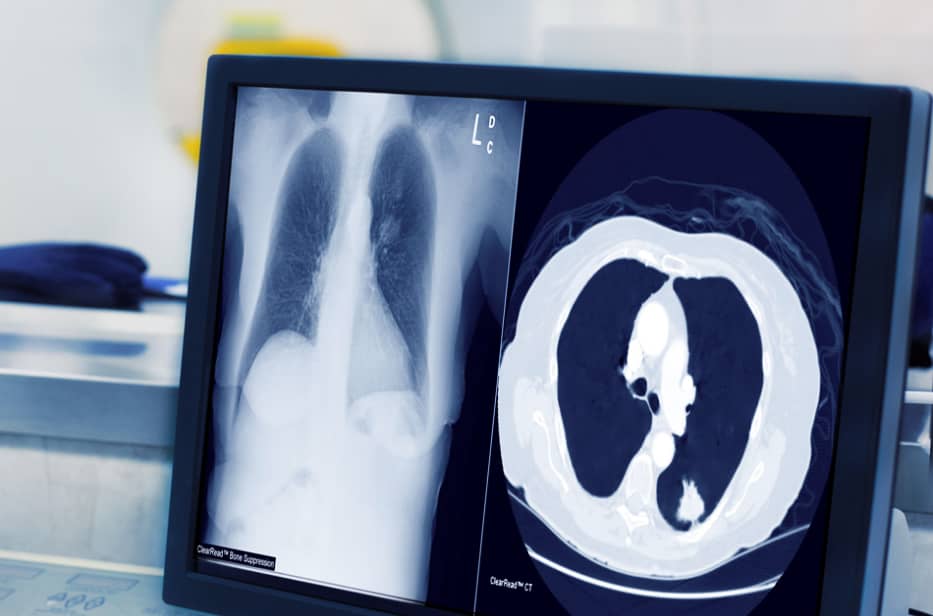
Riverain Technologies advanced AI imaging software is in use by leading healthcare organizations around the world.

Peer reviews, reference accounts, and independent studies confirm that ClearRead technology is positively impacting the field of radiology.
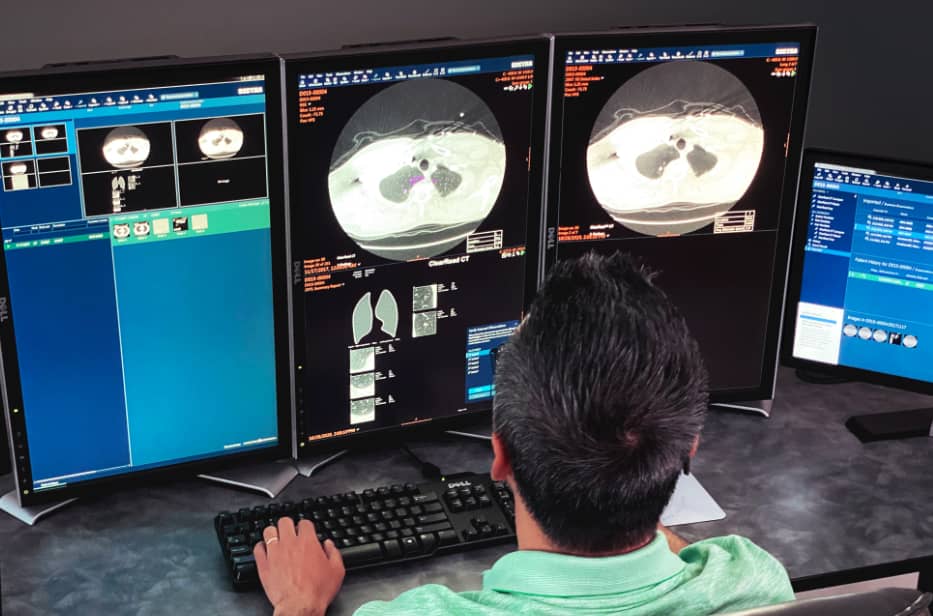
Managing your lung program can seem daunting, whether you’re getting going, growing, or managing workflow. We’ve curated resources for you.
Dayton, Ohio – July 17th, 2018 – ClearRead CTTM is the only FDA-cleared (September 2016) radiology application developed using synthetic data for training a deep learning solution. The concurrent read device aids clinicians in the detection of solid, part solid and ground glass nodules – all early indicators of lung cancer. A pivotal blind multi-reader, multi-case reader study demonstrated that readers using ClearRead CT read 26% faster and detected 29% of previously missed nodules1. Key to achieving the performance was the novel, patent-protected ClearRead CT vessel suppression technology that transforms a standard chest CT into a vessel free series. Results related to the application have been published in the American Journal of Roentgenology1 (March 2018, Volume 210) and in the European Journal of Radiology2 (April 2018, Volume 101).
A fundamental challenge in using artificial intelligence in radiology is the availability of imaging data sets of a scale and precision necessary to optimize machine learning solutions. Riverain has addressed this challenge through the development of synthetic data modeling tools that allow massive training sets to be developed with the necessary supporting data. These advances have led to broad clinical acceptance and accelerating commercial implementation of the company’s software. Revenue in 2018 has grown 94% year-over-year as the company’s tools are increasingly used by radiologists globally in leading health systems such as Duke University, Einstein Medical Center, University of Michigan, University of Maryland, University of Chicago and top Veterans Affairs medical centers.
“Using Riverain’s unique methods, the ClearRead CT software provides proven results, instilling confidence in the clinical decision process,” said Riverain Technologies CEO, Steve Worrell. “Our goal is global widespread adoption and we are well on our way to making a major impact in the Clinical AITM space.”
1. American Journal of Roentgenology: 1-8. 10.2214/AJR.17.18718
2. Vessel suppressed chest Computed Tomography for semi-automated volumetric measurements of solid pulmonary nodules. Milanese, Gianluca et al. European Journal of Radiology, Volume 101, 97 – 102.
Our technology is revolutionizing radiology. We’d love to work with you to help create better outcomes for patients everywhere.
Subscribe to Riverain via Email
Stay up to date on the latest advancements
Riverain Technologies
3130 South Tech Blvd., Miamisburg, OH 45342
800-990-3387
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |