Our technology is revolutionizing radiology. We’d love to work with you to help create better outcomes for patients everywhere.
The first FDA-cleared device that supports concurrent reading, allowing for faster reading with proven, superior, automatic nodule detection.
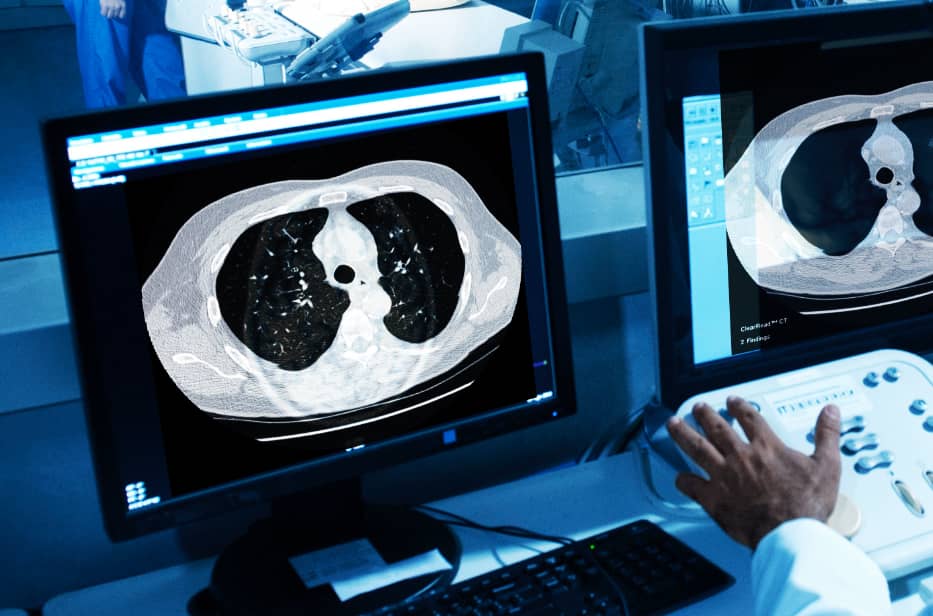
Our five FDA-cleared applications are designed to improve reading efficiency and accuracy across the hospital enterprise without additional equipment, procedures or radiation dose.
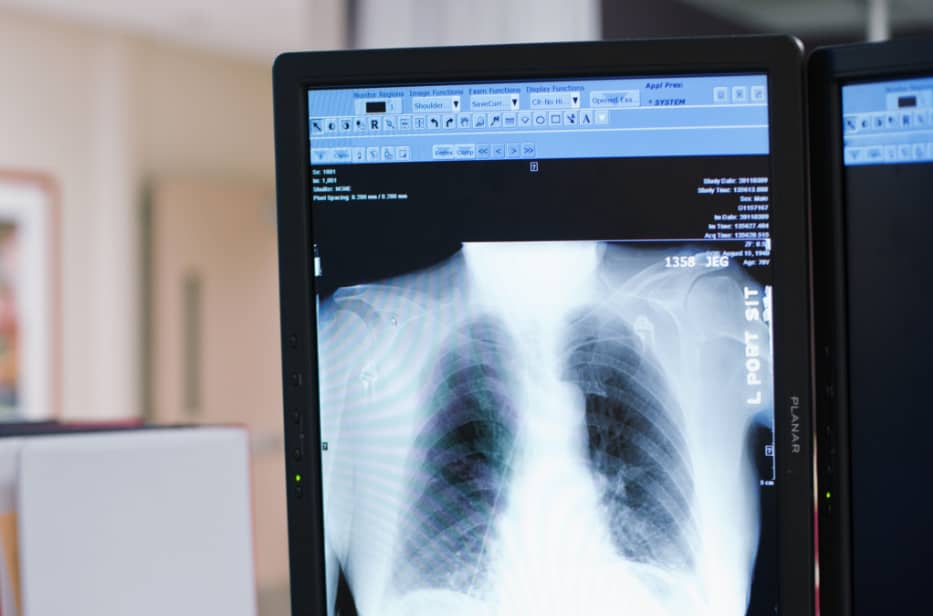
Riverain’s deep learning AI technology is FDA cleared, field tested, and clinically proven to provide real results in radiology.
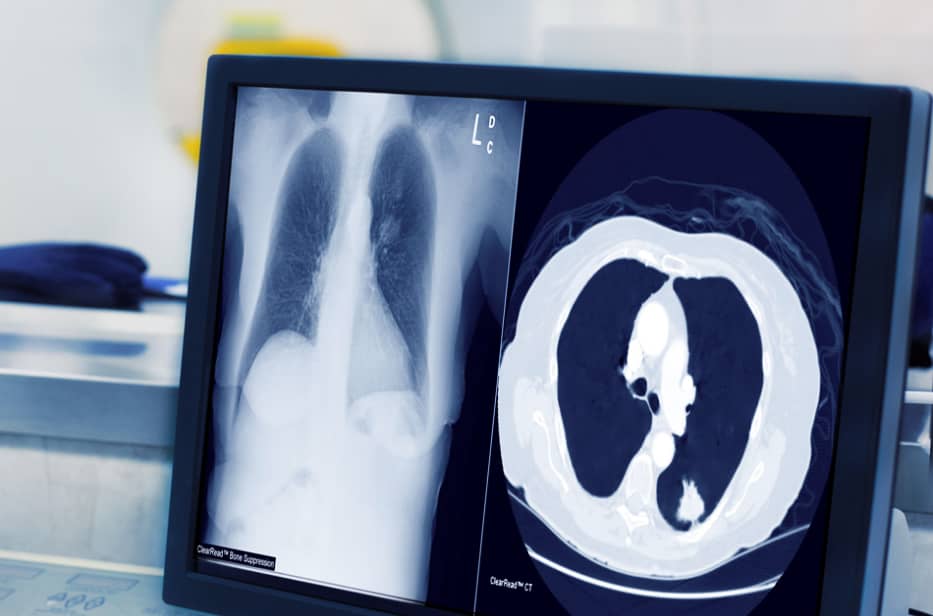
Riverain Technologies advanced AI imaging software is in use by leading healthcare organizations around the world.

Peer reviews, reference accounts, and independent studies confirm that ClearRead technology is positively impacting the field of radiology.
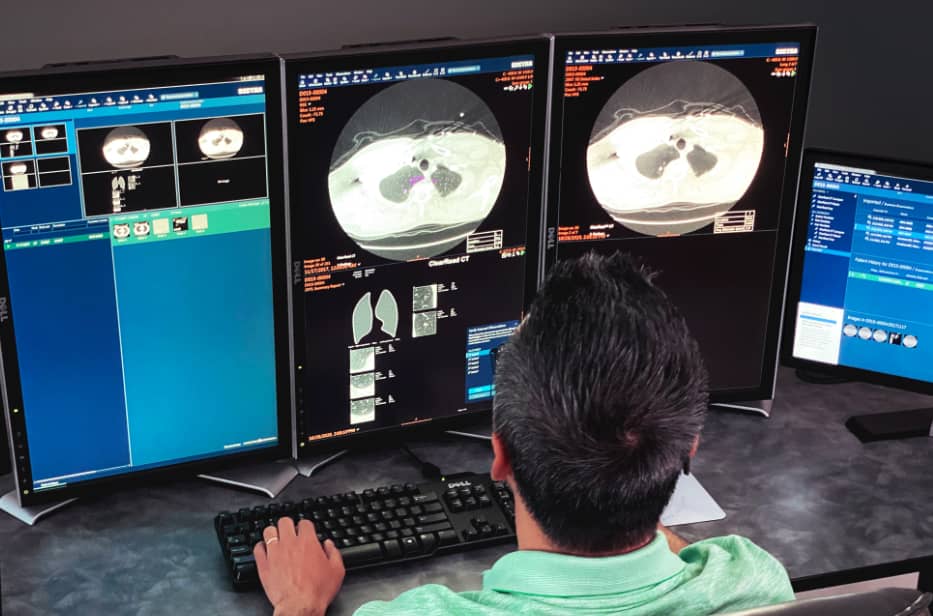
Managing your lung program can seem daunting, whether you’re getting going, growing, or managing workflow. We’ve curated resources for you.
DAYTON, Ohio – April 25, 2017 – Riverain Technologies’ ClearRead CT achieves a significant milestone since the September, 2016 FDA clearance with sales in three continents. Customers are recognizing ClearRead CT as an innovative technology that leads to faster and more accurate detection of actionable lung nodules. The software is the first chest CT tool cleared for the automatic detection of all nodule types, including ground glass, along with being the first system approved for concurrent reading. The solution incorporates Riverain’s patent pending Vessel Suppression technology.
“ClearRead CT has changed the way we read chest CT exams,” said Dr. Terence Matalon, Chairman of Einstein Medical Center Philadelphia’s Department of Radiology. “The application enables identification of small nodules faster and more reliably, particularly for oncological patients.”
Powered by deep learning and acquisition normalization, ClearRead CT automatically and seamlessly processes studies from all CT manufacturers and places the processed series into the existing patient file. Image normalization technology allows ClearRead CT to be rapidly installed and deployed enterprise wide without additional investment in hardware at the facility.
“Installation and implementation of the ClearRead CT software was straightforward, requiring minimal effort from our IT staff,” said Dr. Thomas Frauenfelder, Professor of Radiology at the University Hospital of Zurich. “The ClearRead images are automatically placed into the patient’s file, allowing for quick interpretation. This has proven to be very beneficial in detecting hard to find lung nodules, quickly and efficiently.”
“ClearRead CT allows us to read faster and more accurately. The improved conspicuity of ground glass nodules in the vessel suppressed series is beneficial to both reading speed and accuracy,” said Dr. Kurihara of St. Luke’s International Hospital in Tokyo, Japan. Dr. Matsusako also from St. Luke’s International Hospital, noted, “ClearRead CT is very useful for detecting solid nodules near the hilar area.”
“As CT data volume continues to increase, the ability to improve nodule detection accuracy while maintaining patient volume is increasingly difficult,” said Steve Worrell, CEO of Riverain Technologies. “ClearRead CT is proven to simultaneously improve both nodule detection accuracy and reading efficiency, solving a critical need at a critical time. We are excited to see users embracing ClearRead CT around the world and look forward to working with hospitals and clinicians to improve patient outcomes.”
Our technology is revolutionizing radiology. We’d love to work with you to help create better outcomes for patients everywhere.
Subscribe to Riverain via Email
Stay up to date on the latest advancements
Riverain Technologies
3130 South Tech Blvd., Miamisburg, OH 45342
800-990-3387
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |