Our technology is revolutionizing radiology. We’d love to work with you to help create better outcomes for patients everywhere.
The first FDA-cleared device that supports concurrent reading, allowing for faster reading with proven, superior, automatic nodule detection.
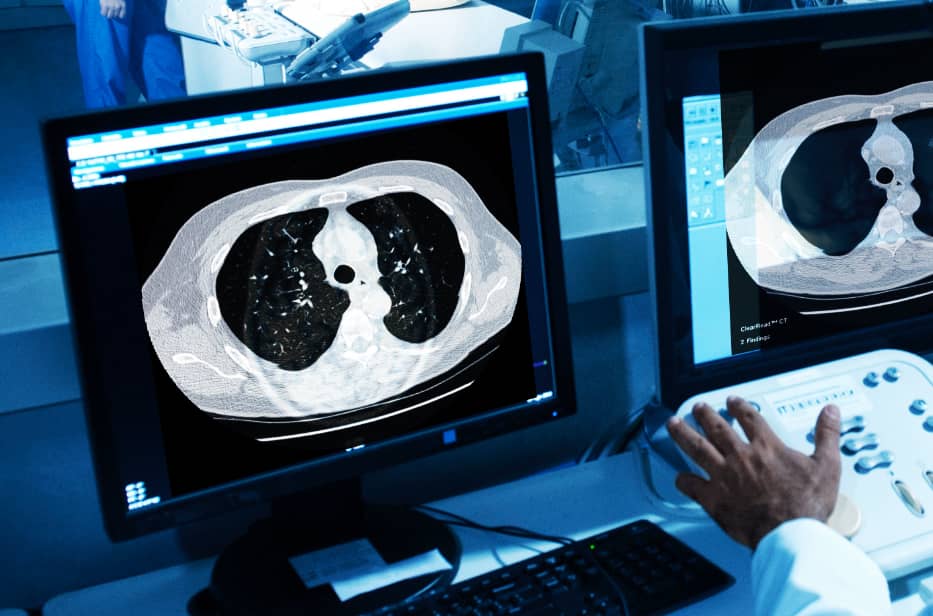
Our five FDA-cleared applications are designed to improve reading efficiency and accuracy across the hospital enterprise without additional equipment, procedures or radiation dose.
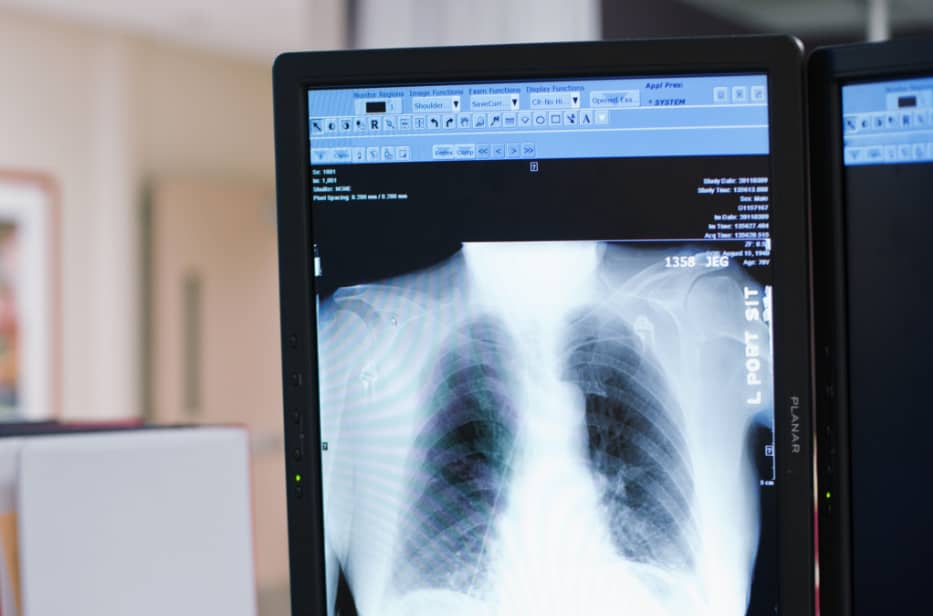
Riverain’s deep learning AI technology is FDA cleared, field tested, and clinically proven to provide real results in radiology.
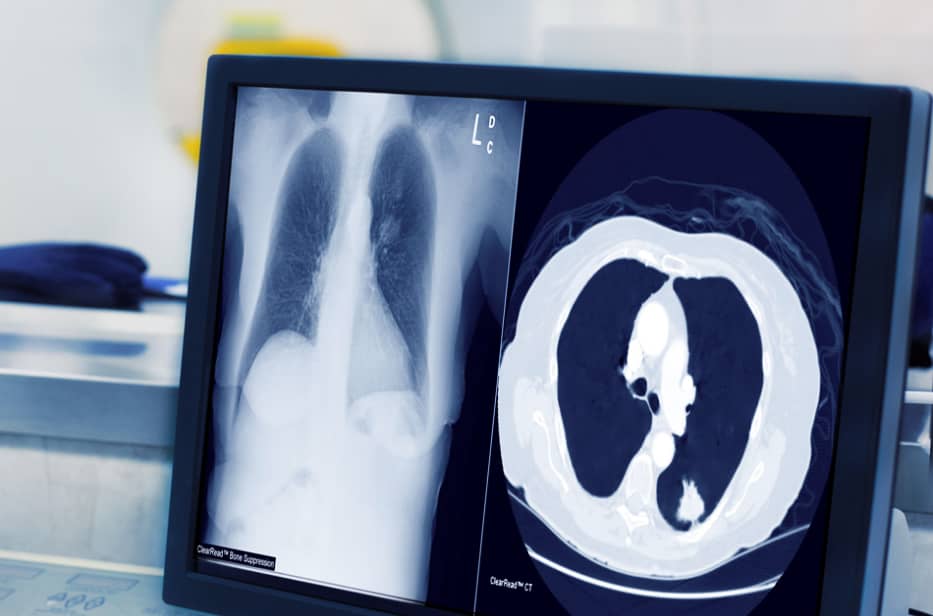
Riverain Technologies advanced AI imaging software is in use by leading healthcare organizations around the world.

Peer reviews, reference accounts, and independent studies confirm that ClearRead technology is positively impacting the field of radiology.
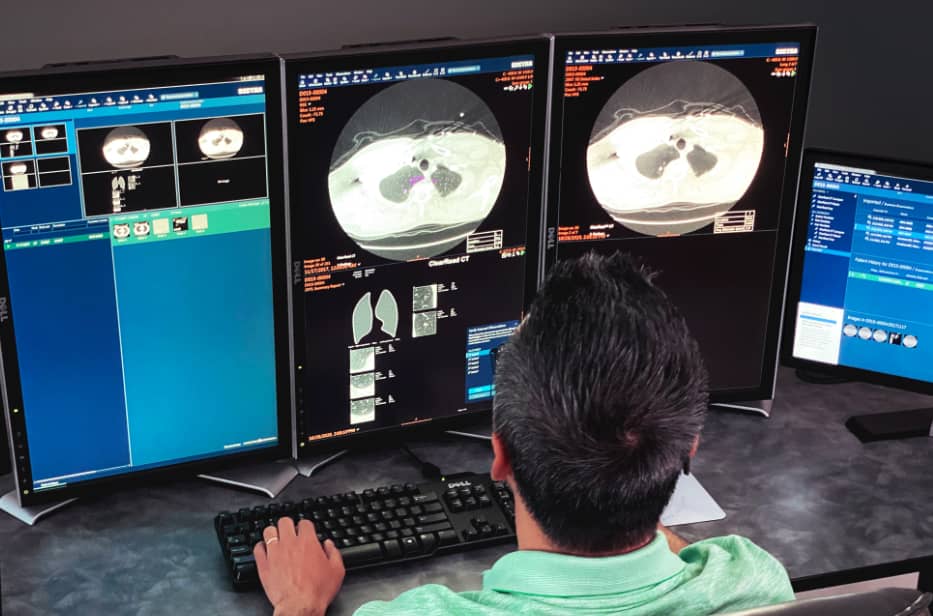
Managing your lung program can seem daunting, whether you’re getting going, growing, or managing workflow. We’ve curated resources for you.
DAYTON, Ohio – June 14, 2017 – With the radiologist’s increasing workload, tools that improve reading efficiency while increasing reading accuracy are imperative. Radiology centers and services are utilizing ClearRead CT as a way to standardize care based on proven reading benefits, ease of use and ease of installation. Leading centers such as, Duke University Medical Center, Saint Luke’s Kansas City, Einstein Medical Center, University of Maryland, and the University of Michigan have deployed ClearRead CT technology along with multiple radiology reading services. As a result, ClearRead CT is now processing data from nearly 50 CT scanners that include all major manufacturer CT devices, while being read on the sites existing picture archiving and communication systems (PACS).
“Riverain’s ClearRead CT has been deployed as a part of our routine Chest CT exams, including patients in our Lung Cancer Screening Program,” said Dr. Jared Christensen, Division Chief of Cardiothoracic Imaging and Imaging Director of Duke’s Lung Cancer Screening Program. “The ClearRead CT technology has helped us to detect lung nodules that may have otherwise been missed. Based upon our early experience, the workflow is faster and more accurate than existing technologies.”
ClearRead CT is the first advanced visualization product cleared by the FDA for concurrent reading, enabling radiologists to read the original series and the processed CT series simultaneously. ClearRead CT is proven to achieve a 26% reduction in reading time and reduce missed nodules by 29%.
ClearRead CT processes CT studies from all manufacturers and automatically places the processed series into the existing patient file prior to the exam review. The seamless integration and ability to process cases from varying acquisition devices and protocols allows the ClearRead CT technology to be deployed enterprise wide, establishing a standard of care for the patient population.
“Using ClearRead CT couldn’t be easier,” said Dr. Charlie White, Professor of Radiology and Vice Chairman of Clinical Affairs. “Image processing is completed in the background so that our normal workflow isn’t interrupted.”
Riverain Technologies has established itself as a dedicated partner to thoracic radiologists and will be exhibiting at the World Congress of Thoracic Imaging (WCTI) in Boston, MA, June 18th – 21st . Visit booth #310 to learn more about the exciting ClearRead CT technology.
Our technology is revolutionizing radiology. We’d love to work with you to help create better outcomes for patients everywhere.
Subscribe to Riverain via Email
Stay up to date on the latest advancements
Riverain Technologies
3130 South Tech Blvd., Miamisburg, OH 45342
800-990-3387
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |