Our technology is revolutionizing radiology. We’d love to work with you to help create better outcomes for patients everywhere.
The first FDA-cleared device that supports concurrent reading, allowing for faster reading with proven, superior, automatic nodule detection.
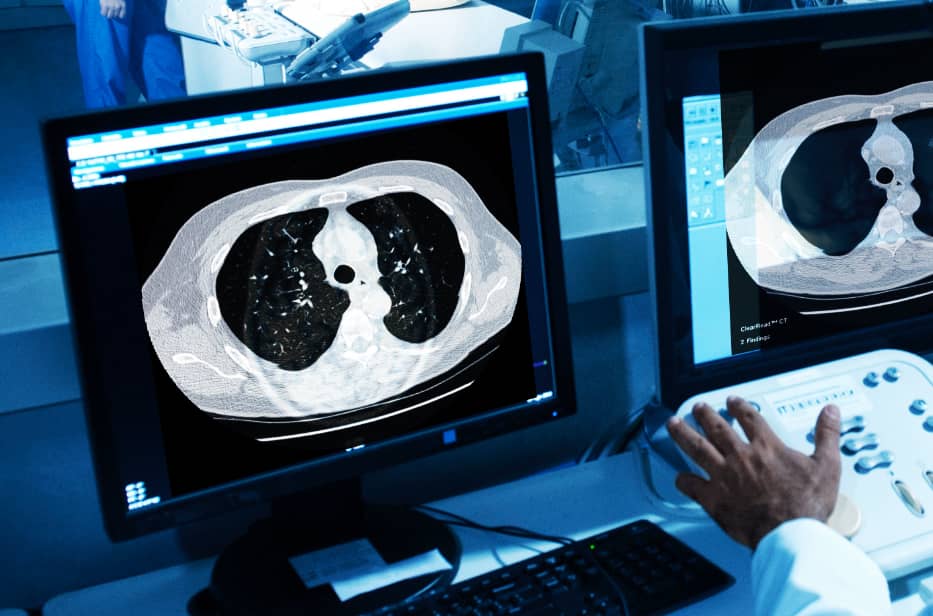
Our five FDA-cleared applications are designed to improve reading efficiency and accuracy across the hospital enterprise without additional equipment, procedures or radiation dose.
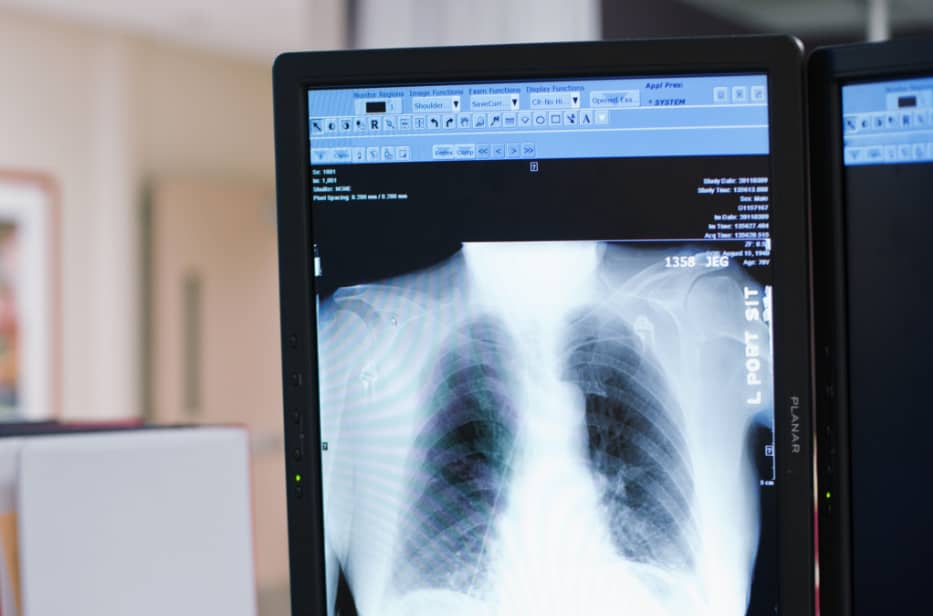
Riverain’s deep learning AI technology is FDA cleared, field tested, and clinically proven to provide real results in radiology.
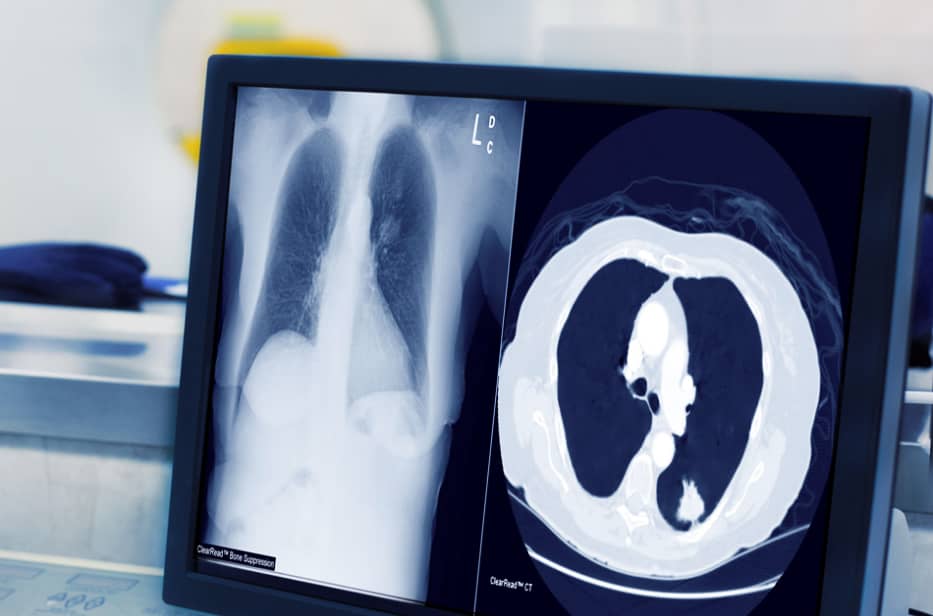
Riverain Technologies advanced AI imaging software is in use by leading healthcare organizations around the world.

Peer reviews, reference accounts, and independent studies confirm that ClearRead technology is positively impacting the field of radiology.
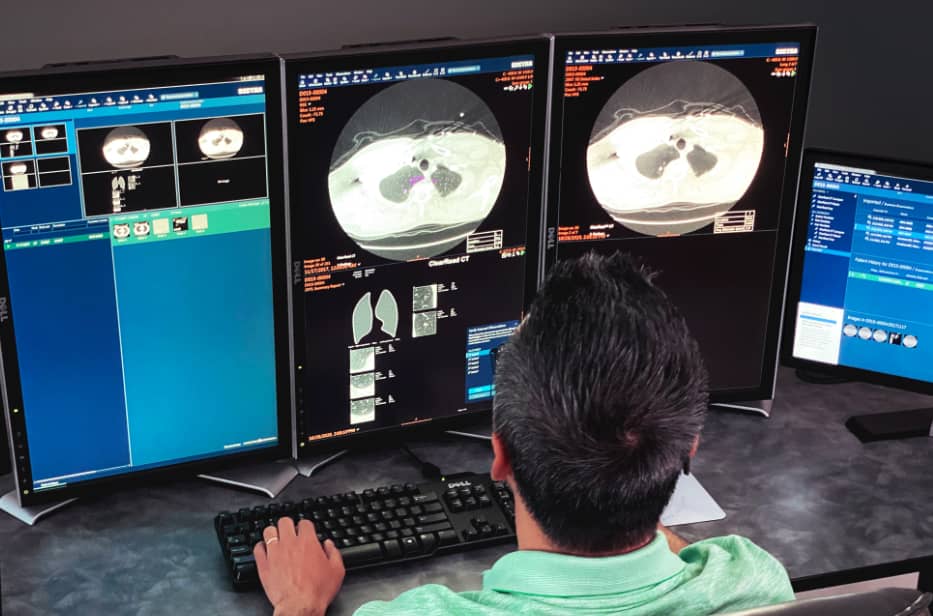
Managing your lung program can seem daunting, whether you’re getting going, growing, or managing workflow. We’ve curated resources for you.
DAYTON, Ohio – November 21, 2017 – Riverain Technologies announces that it has integrated its ClearRead CT reporting with the Nuance PowerScribe 360 reporting and visualization tools. As Riverain’s ClearRead CT install base continues to expand and customers implement enterprise level solutions, tighter integration into reporting tools is critical for optimized workflows.
ClearRead CT is the first advanced visualization product cleared by the FDA for concurrent reading, enabling radiologists to read the original and the processed CT series simultaneously. ClearRead CT is proven to achieve a 26% reduction in reading time and reduce missed nodules by 29%. The technology processes CT studies from all manufacturers and automatically places the processed series into the existing patient file. The seamless integration and ability to process cases from varying acquisition devices and protocols allows the ClearRead CT technology to be deployed enterprise wide, enabling a standard of care for the patient population.
“The integration of ClearRead CT and Nuance PowerScribe 360 reporting and visualization tools provides another critical step in ensuring ClearRead CT achieves optimal clinical performance,” said Steve Worrell, CEO of Riverain Technologies. “Riverain is excited to be working with Nuance based on its leadership in clinical workflow solutions.”
“Nuance Communications is the leader in providing automated reporting and visualization tools, and Nuance PowerScribe 360 has become the gold standard in clinical reporting. We are pleased to work with Riverain Technologies as they continue to build enterprise solutions that improve the early detection of lung disease,” said Karen Holzberger, VP and GM, Healthcare Diagnostics, Nuance Communications.
Riverain Technologies has established itself as a dedicated partner to thoracic radiologists and will be exhibiting at the Radiological Society of North America (RSNA) conference in Chicago, IL, November 26th – 30th. Visit booth #3710 or #8149P in the Machine Learning Showcase to learn more about the ClearRead CT technology.
Our technology is revolutionizing radiology. We’d love to work with you to help create better outcomes for patients everywhere.
Subscribe to Riverain via Email
Stay up to date on the latest advancements
Riverain Technologies
3130 South Tech Blvd., Miamisburg, OH 45342
800-990-3387
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |