Our technology is revolutionizing radiology. We’d love to work with you to help create better outcomes for patients everywhere.
The first FDA-cleared device that supports concurrent reading, allowing for faster reading with proven, superior, automatic nodule detection.
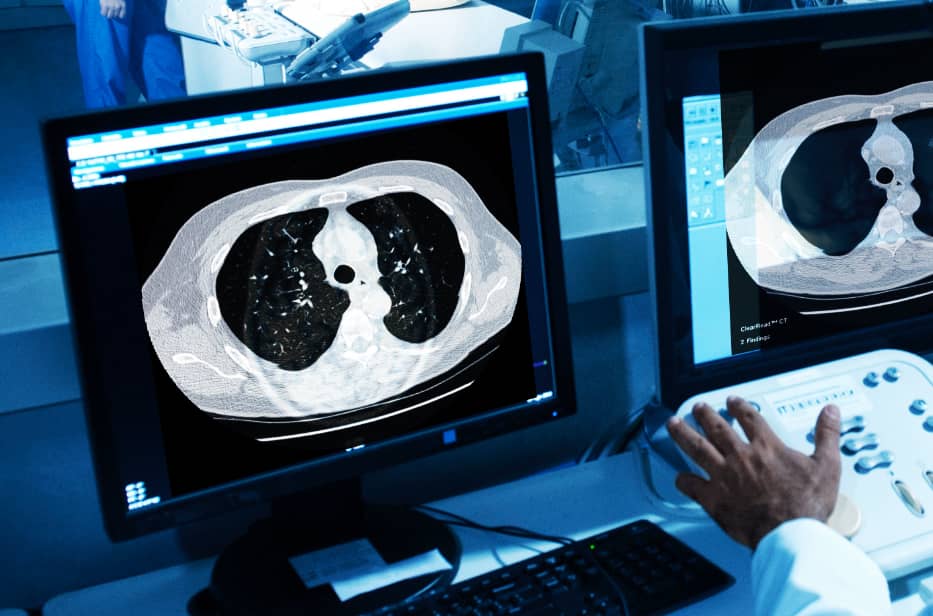
Our five FDA-cleared applications are designed to improve reading efficiency and accuracy across the hospital enterprise without additional equipment, procedures or radiation dose.
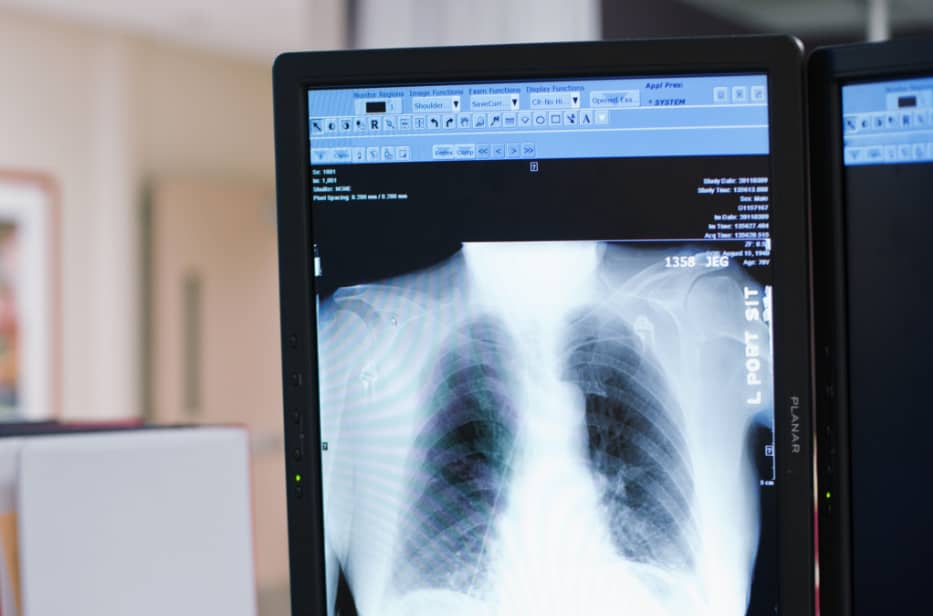
Riverain’s deep learning AI technology is FDA cleared, field tested, and clinically proven to provide real results in radiology.
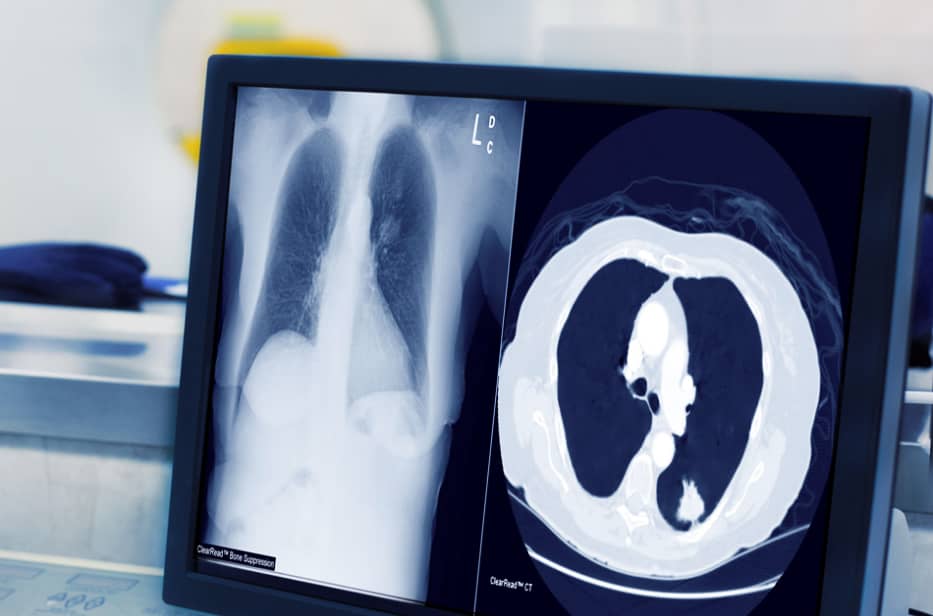
Riverain Technologies advanced AI imaging software is in use by leading healthcare organizations around the world.

Peer reviews, reference accounts, and independent studies confirm that ClearRead technology is positively impacting the field of radiology.
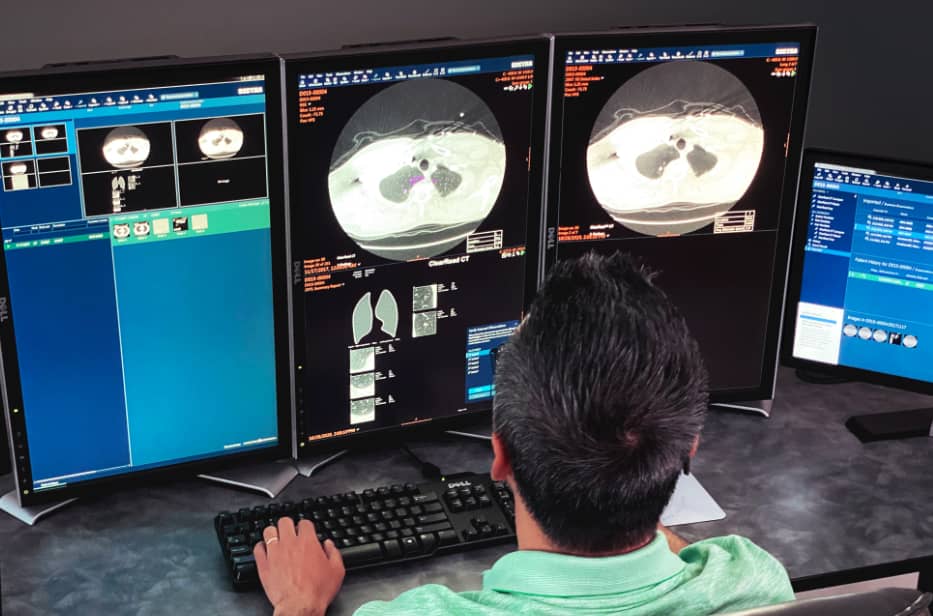
Managing your lung program can seem daunting, whether you’re getting going, growing, or managing workflow. We’ve curated resources for you.
DAYTON, Ohio – September 19, 2016 – Riverain Technologies is committed to providing cutting edge software to aid the clinician in the efficient, effective early detection of lung disease. Today, Riverain Technologies announces ClearRead CT, a breakthrough nodule detection application, has received clearance from the Food and Drug Administration (FDA).
ClearRead CT is the only FDA cleared device to support concurrent reading, allowing for faster reading with proven superior automatic nodule detection performance for all primary nodule types, including: solid, sub-solid and ground glass nodules.
ClearRead CT, powered by acquisition normalization technology, provides an enterprise ready solution for the entire healthcare network. The technology seamlessly processes CT studies from a wide range of manufacturers and acquisition protocols and quickly installs into the clinical environment without the need for new hardware or customized tuning to specific devices or protocols.
ClearRead CT was evaluated in a multi-reader, multi-case clinical trial and showed radiologists achieved a 29% reduction in missed actionable nodules, while reducing reading time by 26%.
ClearRead CT is comprised of two powerful tools, ClearRead CT | Vessel Suppress and ClearRead CT | Detect. Deep learning enables the vessel suppression technology to assist both machine and humans in the detection and characterization of all primary nodule types, allowing for previously unattained nodule detection performance.
“Achieving high reading efficiency while maintaining accuracy is increasingly important given the burden placed on today’s radiologists. With more patients and increased data volume due to thinner sections, ClearRead CT offers a critical advantage.” said Steve Worrell, Riverain’s Chief Executive Officer. “The use of deep learning and other key technologies to suppress the vessels in CT studies proved to be essential in realizing a product capable of achieving the two competing objectives.”
ClearRead CT is available for sale in the United States, Canada and Europe.
Our technology is revolutionizing radiology. We’d love to work with you to help create better outcomes for patients everywhere.
Subscribe to Riverain via Email
Stay up to date on the latest advancements
Riverain Technologies
3130 South Tech Blvd., Miamisburg, OH 45342
800-990-3387
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |